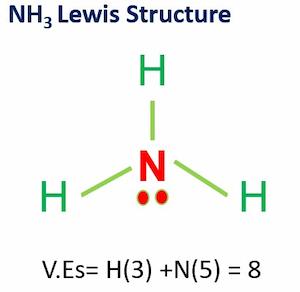
Ammonium dihydrogen phosphate (ADP) is the ammonium salt of phosphoric acid (molar ratio 1:1). It is used as a fertilizer. It contains dihydrogen phosphate. It is also known as monoammonium phosphate (MAP). ADP is a key component in agricultural fertilizers as well as several fire extinguishers.
It is also used extensively in optics and electronics.
| Compound Name | Ammonium dihydrogen phosphate |
| Sulfurous acid formula | (NH4)H2PO4 |
| Color/Form | White, tetrahedral crystals |
| Density | 1.80 g/cu cm |
| Molecular Weight | 115.03 g/mol |
Table of Contents
Ammonium dihydrogen phosphate- Key Points
- Decomposes to ammonia and phosphoric acid when heated.
- pH of 0.2 Molar aq soln: 4.2
- slightly soluble in alcohol; practically insoluble in acetone
- Stable in air
- Used in dry chemical fire extinguishers
Preparation
Ammonium dihydrogen phosphate (ADP) is manufactured industrially by the exothermic reaction of phosphoric acid and ammonia in the following proportions:
NH3 + H3PO4 → NH6PO4
Crystalline ADP then precipitates.
Related Links
Latest posts by Umair Javaid, PhD Student (see all)
- BCl3 Lewis Structure in four simple steps - November 1, 2023
- PH3 Lewis Structure in four simple steps - October 8, 2023
- PF3 Lewis structure in four simple steps - September 24, 2023