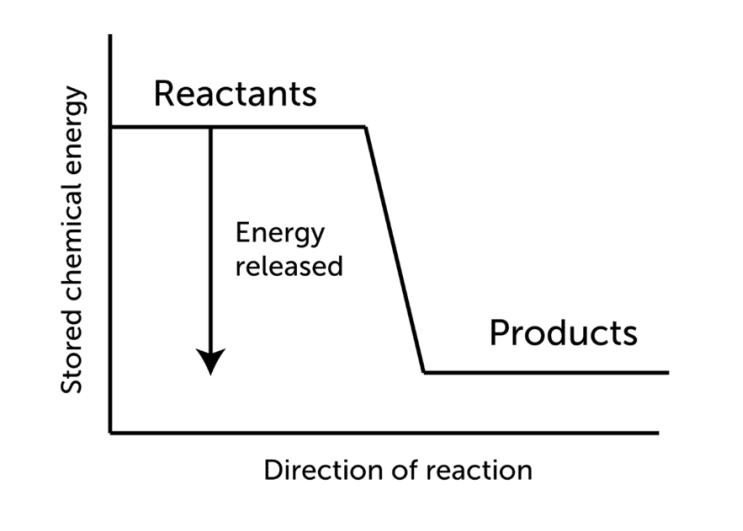
Fluoroantimonic acid (HSbF6), is the world’s most powerful superacid. It’s made by combining hydrogen fluoride (HF) with antimony pentafluoride (SbF5). The superacid is produced by many combinations, but the strongest superacid known to man is produced by combining equal quantities of the two acids.

Table of Contents
Fluoroantimonic Acid- Important Points
- The reaction of hydrogen fluoride (HF) and SbF5 is exothermic.
- The octahedral SbF6 is formed when HF releases its proton (H+) and its conjugate base (F) is sequestered by one or more molecules of SbF5.
- This anion is classified as noncoordinating because it is both a very weak nucleophile and a very weak base.
- The proton effectively becomes “naked,” explaining the system’s high acidity.
- Fluoroantimonic acid is 21019 times more powerful than pure sulfuric acid.
- Superacids are commonly used to produce and retain organic cations, which are important as intermediate intermediates in a variety of processes, including the manufacture and research of plastics and high-octane gasoline.
Related Links
N2O | Laughing Gas
HCN Lewis Structure| Step By Step Construction
SO2 (Sulfur Dioxide)
SiO2 Lewis Structure| Step By Step Construction
Hydrogen Bond| Definition and Easy Explanation
Latest posts by Umair Javaid, PhD Student (see all)
- BCl3 Lewis Structure in four simple steps - November 1, 2023
- PH3 Lewis Structure in four simple steps - October 8, 2023
- PF3 Lewis structure in four simple steps - September 24, 2023