A binary compound is something consisting of precisely two elements.
An element is a type of substance that can’t be further divided into simpler substances through chemical methods. Any of those boxes on a periodic table of elements represent an element. A few examples of chemical elements are hydrogen, oxygen, and iron.
This means that a binary compound will be composed of two different chemical elements.
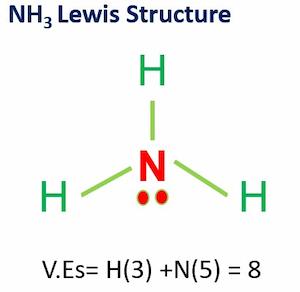
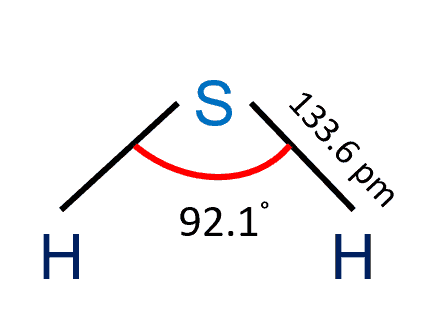
Water (H2O), Hydrogen sulfide( H2S), and Ammonia (NH3) are examples of binary compounds.
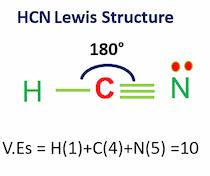
Gold (Au), Iron(Fe), Hydrogen cyanide (HCN), and Nitric acid (HNO3) are examples of substances that are not chemical compounds.
More Interesting Topics
H2S Lewis Structure & Molecular Geometry
O3 Lewis Structure | Step By Step Drawing
H2S Polar Or Nonpolar
Sulfurous Acid| Formula & Lewis Structure
Sodium Sulfate- Miraculous salt|
CH4 Polarity
Hydrogen Cyanide-A Simple Overview
Hydrogen Electronegativity
Latest posts by Umair Javaid, PhD Student (see all)
- BCl3 Lewis Structure in four simple steps - November 1, 2023
- PH3 Lewis Structure in four simple steps - October 8, 2023
- PF3 Lewis structure in four simple steps - September 24, 2023