N2O molecular geometry is a linear shape with a bond angle of 180 degrees.
Nitrogen(N) and Oxygen(O) atoms are covalently linked in the N2O molecular geometry. N and O have five and six valence electrons, respectively. N2O contains a total of 16 valence electrons. N2O, also known as nitrous oxide, is frequently referred to as laughing gas. There are numerous additional names for this chemical, such as sweet air, protoxide of nitrogen, and so on.
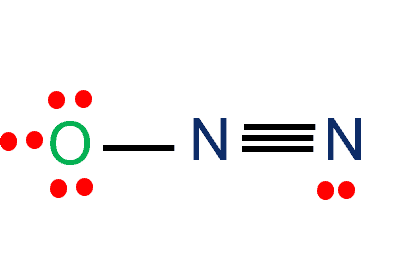
Table of Contents
What is Nitrous Oxide?
Nitrous oxide (N2O) is useful in a number of medicinal applications due to its anaesthetic properties. It is insoluble in water and, at higher temperatures, acts as a strong oxidant. It has a somewhat pleasant odour and is colourless. At greater doses, it can have a narcotic effect.
| Name of molecule | Nitrous oxide (N2O) |
| Bond Angles | 180 degrees |
| N2O molecular geometry | Linear |
| No of Valence Electrons in the molecule | 16 |
| The dipole moment of N2O | 0.160 D [Reference] |
N2O Lewis Structure
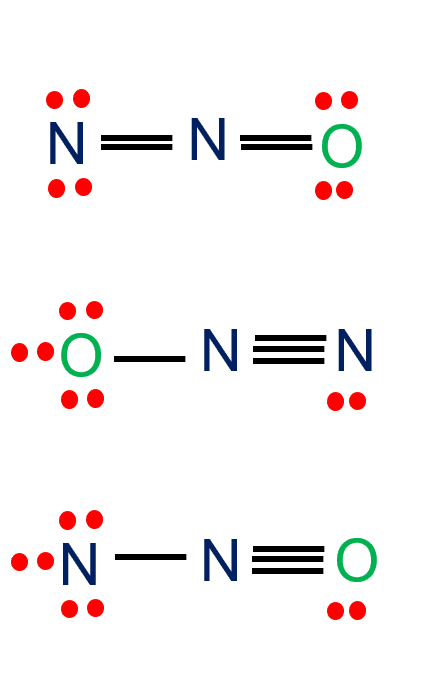
The total Nitrogen to Oxygen atom ratio in the N2O Lewis Structure is 2:1. Three possible Lewis structures OF N2O are given in the figure below. For details, check the full article about N2O lewis structure.
Oxidation number of NH3
Ammonia is made up of one nitrogen atom and three hydrogen atoms (NH3). The NH3 oxidation number is zero because the sum of the individual oxidation values of the atoms nitrogen (oxidation number = -3) and hydrogen (oxidation number = 1) is zero.
Check another interesting topic “Is NH3 a polar molecule?”.
Related Links
CO2 Lewis Structure and Molecular Geometry
HCN Lewis Structure| Step By Step Construction
SiO2 Lewis Structure| Step By Step Construction
SO2 (Sulfur Dioxide) Lewis structure
NH3 Lewis Structure & Molecular Geometry
O2 Lewis Structure & Molecular Geometry
CH4 Lewis Structure & Molecular Geometry
Is Nh3 Polar?
- BCl3 Lewis Structure in four simple steps - November 1, 2023
- PH3 Lewis Structure in four simple steps - October 8, 2023
- PF3 Lewis structure in four simple steps - September 24, 2023