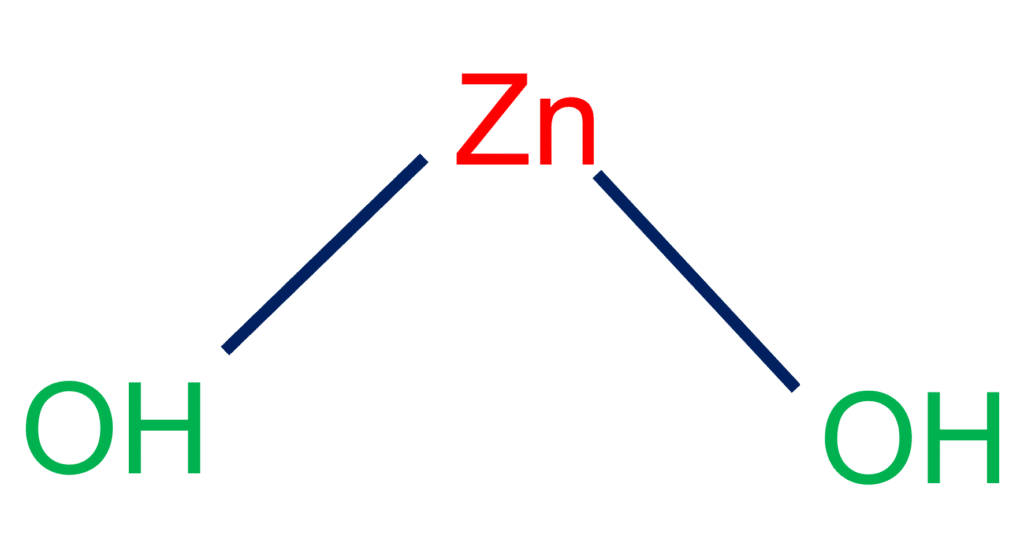
Zinc hydroxide (Zn(OH)2) is an inorganic chemical compound found in nature as a rare mineral. It is classified as an amphoteric chemical because it dissolves equally in strong acid and basic solutions. Zn(OH)2 dissociates into zinc and hydroxide ions when exposed to water.
| Compound Name | Zinc hydroxide- (Zn(OH)2 |
| Appearance | White powder |
| Melting point | 125° C (257° F) |
| Molar Mass | 99.424 g/mol |
| Density | 3.05 g/cm³ |
| Solubility | The zinc hydroxide is slightly soluble in water and insoluble in alcohol. |
An amphoteric compound can act as either an acid or base.
In addition, An amphoteric substance has transferable hydrogen and an atom with lone electron pairs.
Some examples of amphoteric chemical substances are amino acids, water, proteins, and metal oxides.
Table of Contents
What is Zinc hydroxide- (Zn(OH)2?
Zinc hydroxide, like the hydroxides of other metals such as lead, aluminum, beryllium, tin, and chromium, is amphoteric. As a result, it dissolves easily in alkaline solutions like sodium hydroxide as well as dilute solutions of strong acids like HCl.
Zinc hydroxide is a rare natural mineral found on Earth. It is also an amphoteric white solid that is soluble in strong acid or basic solutions. It may be found in three rare earth minerals: ashoverite, wulfenite, and others.
Zinc hydroxide- (Zn(OH)2 ionic or covalent?
Zinc hydroxide contains a metal (i.e. zinc). Hence, it is an ionic compound.
What are Zinc and Hydoroxde Compounds?
Zinc has the chemical symbol Zn and the atomic number 30. At room temperature, zinc is a somewhat brittle metal that turns silvery-gray when deoxidized. It is the first element in periodic table group 12 (IIB).
Hydroxide is any substance comprising one or more groups, each containing one atom that is coupled with oxygen and hydrogen to form a negatively charged ion.
Related Links
Combustion Reactions| Introduction, Reaction, & Facts
Is NH3 Acid or Base?| NH3 Properties
Acidic Hydrogen
Conduction in Physics| Easy Examples
Sodium Phosphate – Formula, Structure, Types, and Uses
How cold is Liquid Nitrogen?
Frequently Asked Questions
1. Is chlorine a metal?
Chlorine (Cl) is the second lightest member of the periodic table’s halogen elements, or Group 17 (Group VIIa). Because it lacks metal-like properties such as electrical conductivity, flexibility, and strength, chlorine is classified as a nonmetal.
Check the full article “Is chlorine a metal?”.
2. Is hydrogen a metal?
While hydrogen has the same ns1 electron configuration as alkali metals, it is not considered a metal because it forms cations (H+) more slowly than other alkali metals. As a result, the simple answer to the question “is hydrogen a metal?” is no.
3. What is magnesium chloride?
Magnesium Chloride (MgCl2) is an ionic chemical and magnesium source that is used to replace electrolytes and cure magnesium shortage diseases. Magnesium Chloride anhydrous contains 25.5 percent elemental magnesium by mass.
4. What is sulfur electronic configuration?
Sulfur electronic configuration is 1s2 2s2 2p6 3s2 3p4
5. What is Zinc Hydroxide?
Zinc hydroxide is an inorganic chemical compound with an amphoteric character and may dissolve in both a strong basic and an acid solution. It is frequently used as a galvanic material to prevent metal substrate corrosion. The zinc hydroxide formula is Zn(OH)2.
More Links
Is Water a Mixture?
How Many Cups in a Gallon? Cups to Pints, Quarts, and More
How cold is Liquid Nitrogen?
Viscosity of Water
The pH of Distilled/ De-Ionized Water
SiCl4 Polar or Nonpolar| Easy Explanation
- BCl3 Lewis Structure in four simple steps - November 1, 2023
- PH3 Lewis Structure in four simple steps - October 8, 2023
- PF3 Lewis structure in four simple steps - September 24, 2023