The ozone layer is a thin layer of the Earth’s atmosphere that absorbs the majority of the sun’s UV energy. When it is depleted, more UV radiation reaches the surface, potentially harming humans and other living things.
Table of Contents
What is the Ozone Layer?
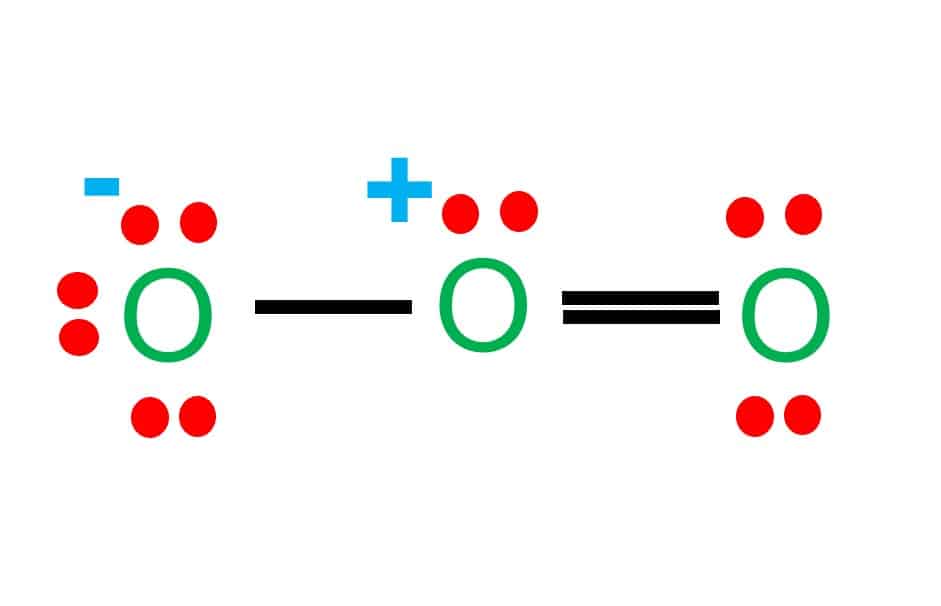
Ozone, often known as O3, is a molecule composed of three oxygen atoms. When heat and sunshine trigger a chemical reaction between nitrogen oxides (NOX) and volatile organic compounds (VOC), commonly known as hydrocarbons, ozone is produced.
Typically, the ozone layer absorbs 97–99 percent of incoming UV-B light. Higher ozone concentrations in the stratosphere are therefore critical to ensuring that life (including humans) on Earth’s surface is not exposed to dangerous UV-B radiation concentrations.
The quantity of ozone over a given area on Earth changes naturally with latitude, season, and day of the week. The ozone layer is strongest at the poles and weakest around the equator under typical conditions.
Ozone Layer Depletion
Ozone layer depletion is the weakening of the upper atmosphere’s ozone layer. This occurs when chlorine and bromine atoms in the atmosphere come into contact with ozone and destroy it.
When chlorine and bromine atoms come into contact with ozone molecules in the stratosphere, they destroy them. One chlorine atom may damage over 100,000 ozone molecules before it is evacuated from the stratosphere. Ozone may be destroyed faster than it can be generated.
When certain chemicals are exposed to intense UV radiation, they emit chlorine and bromine, which leads to ozone layer depletion. These substances are known as Ozone Depleting Substances (ODS).
More Links
- BCl3 Lewis Structure in four simple steps - November 1, 2023
- PH3 Lewis Structure in four simple steps - October 8, 2023
- PF3 Lewis structure in four simple steps - September 24, 2023